WHO GMP Certified
US FDA Compliant
ISO 14644 Class 100
Leading Manufacturer of
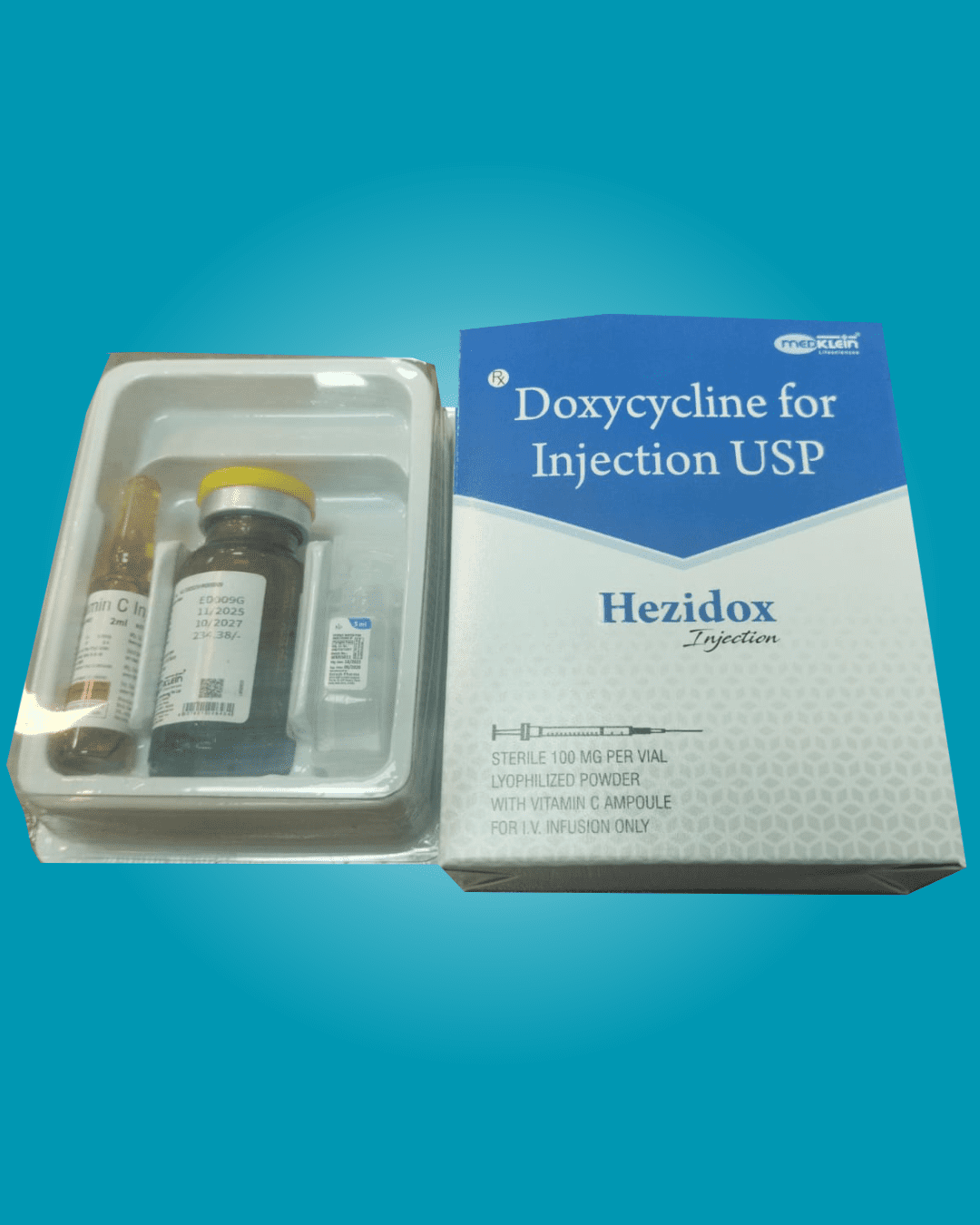
Dry Injections
Liquid Injections
IV Fluids
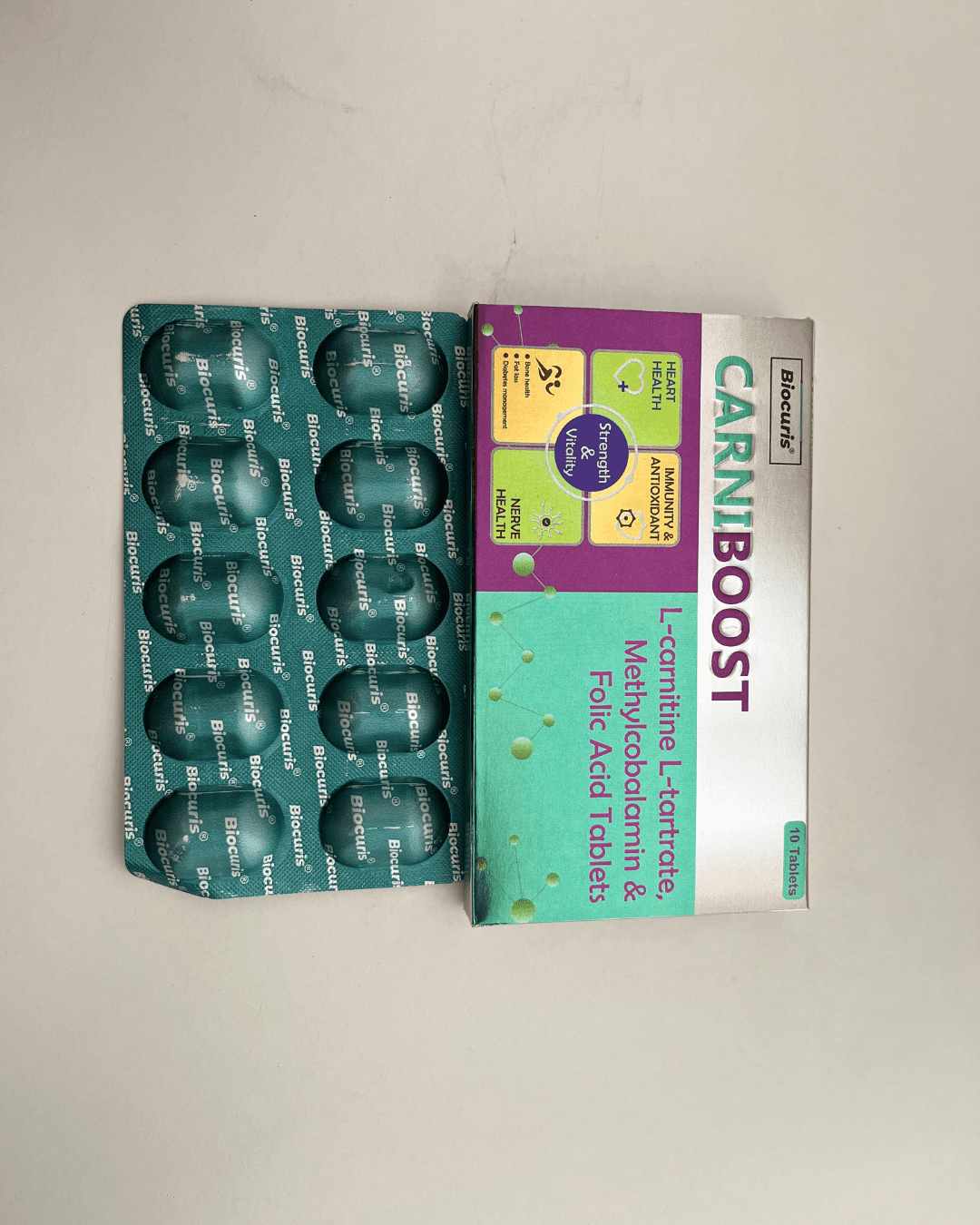
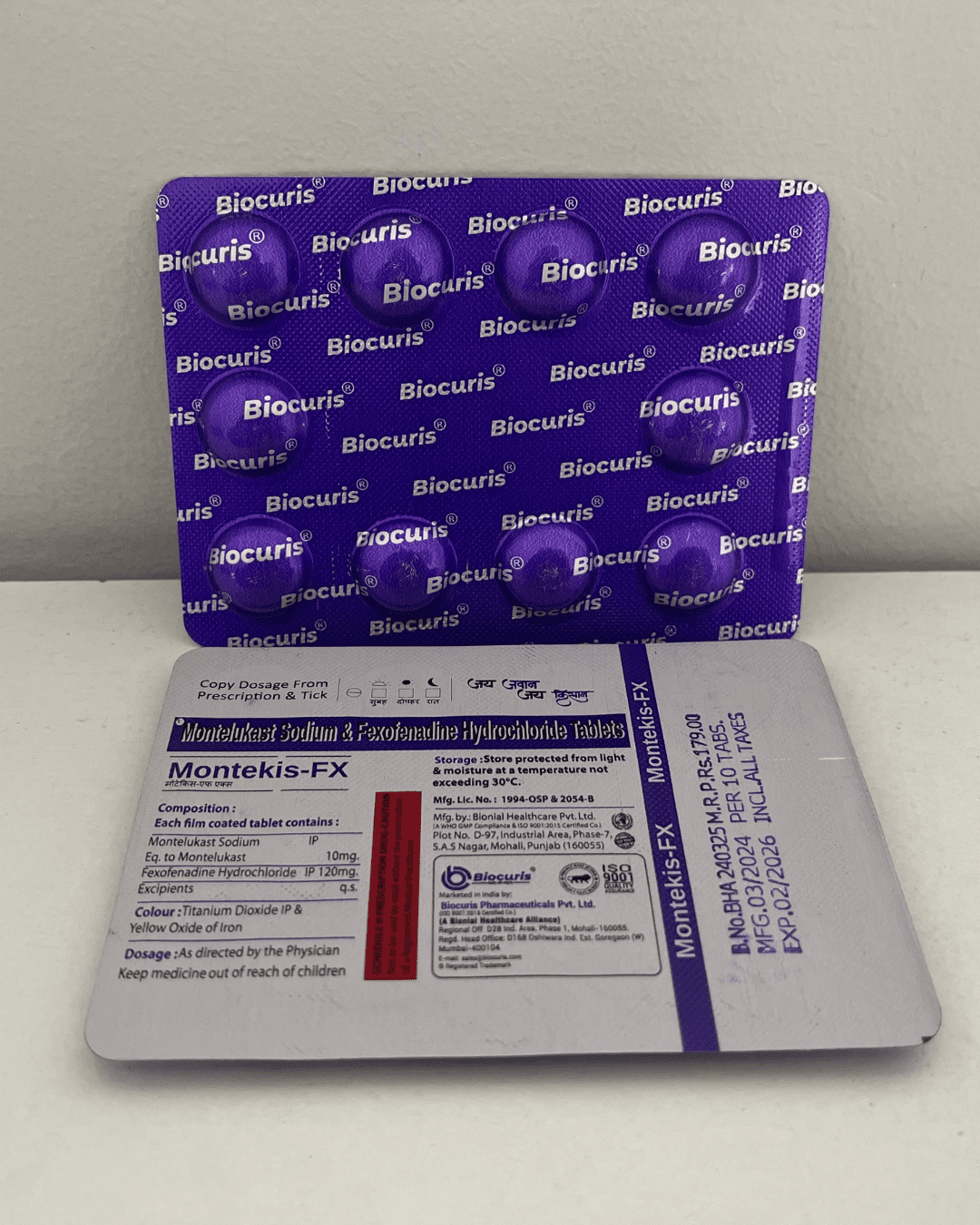
Tablets
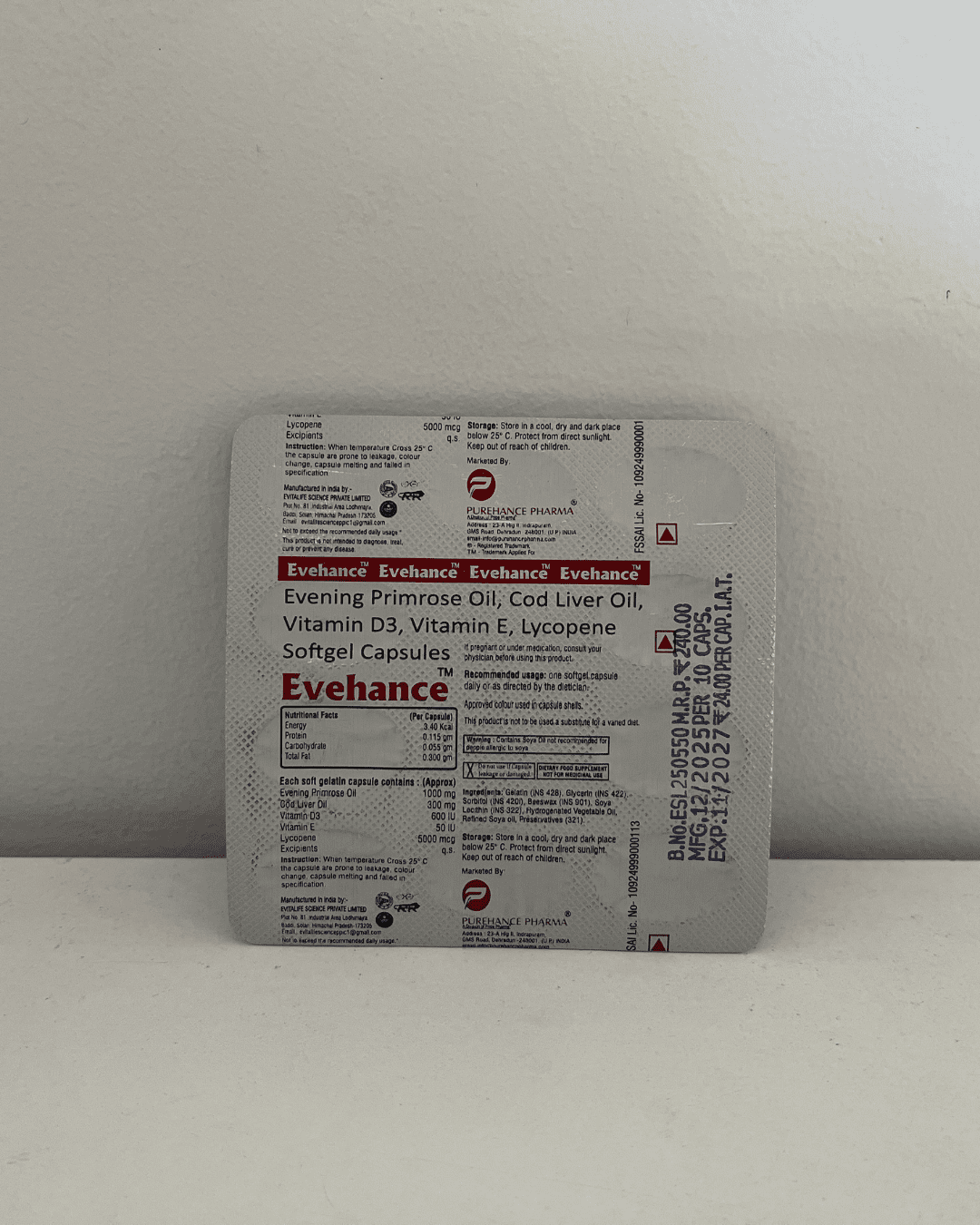
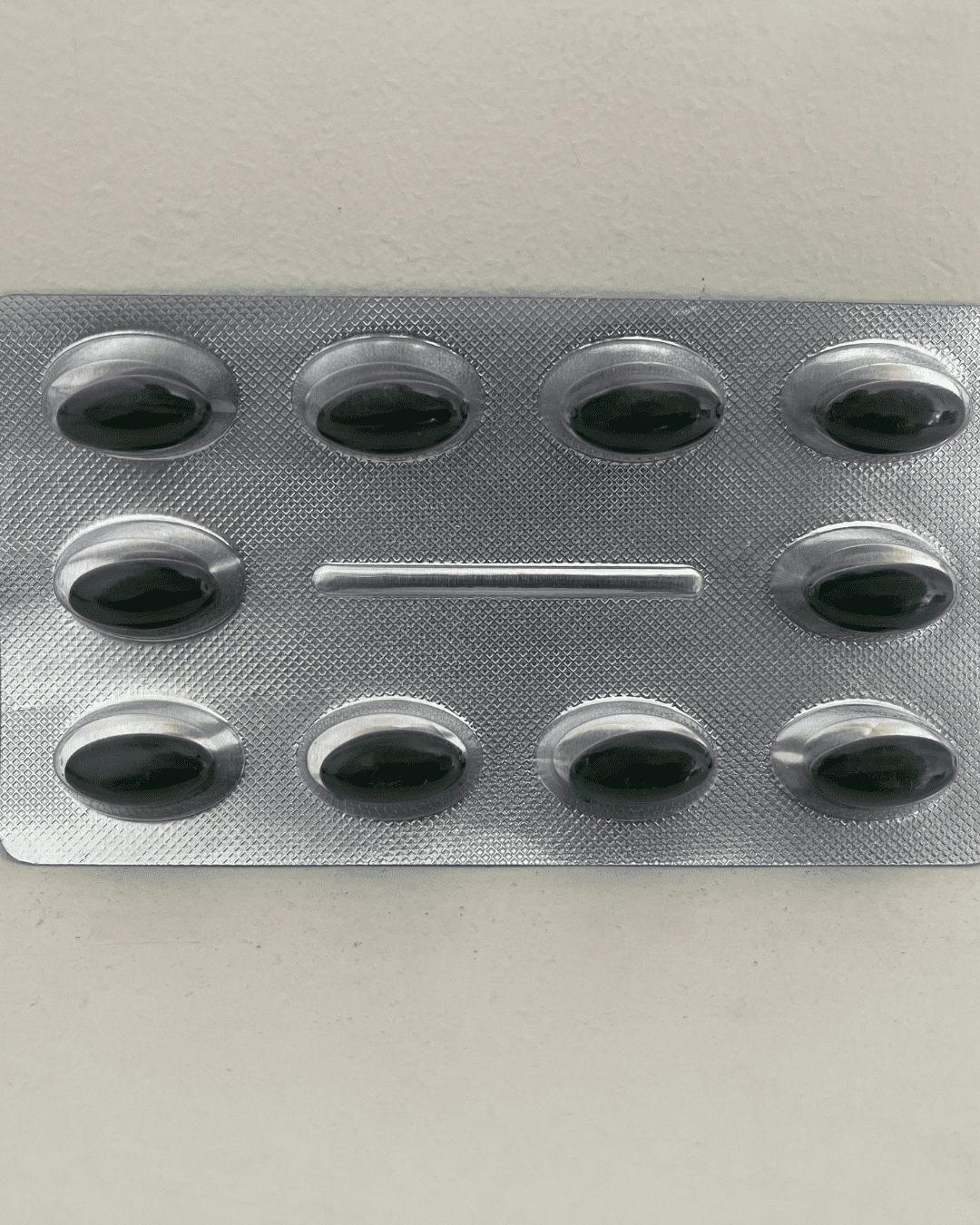
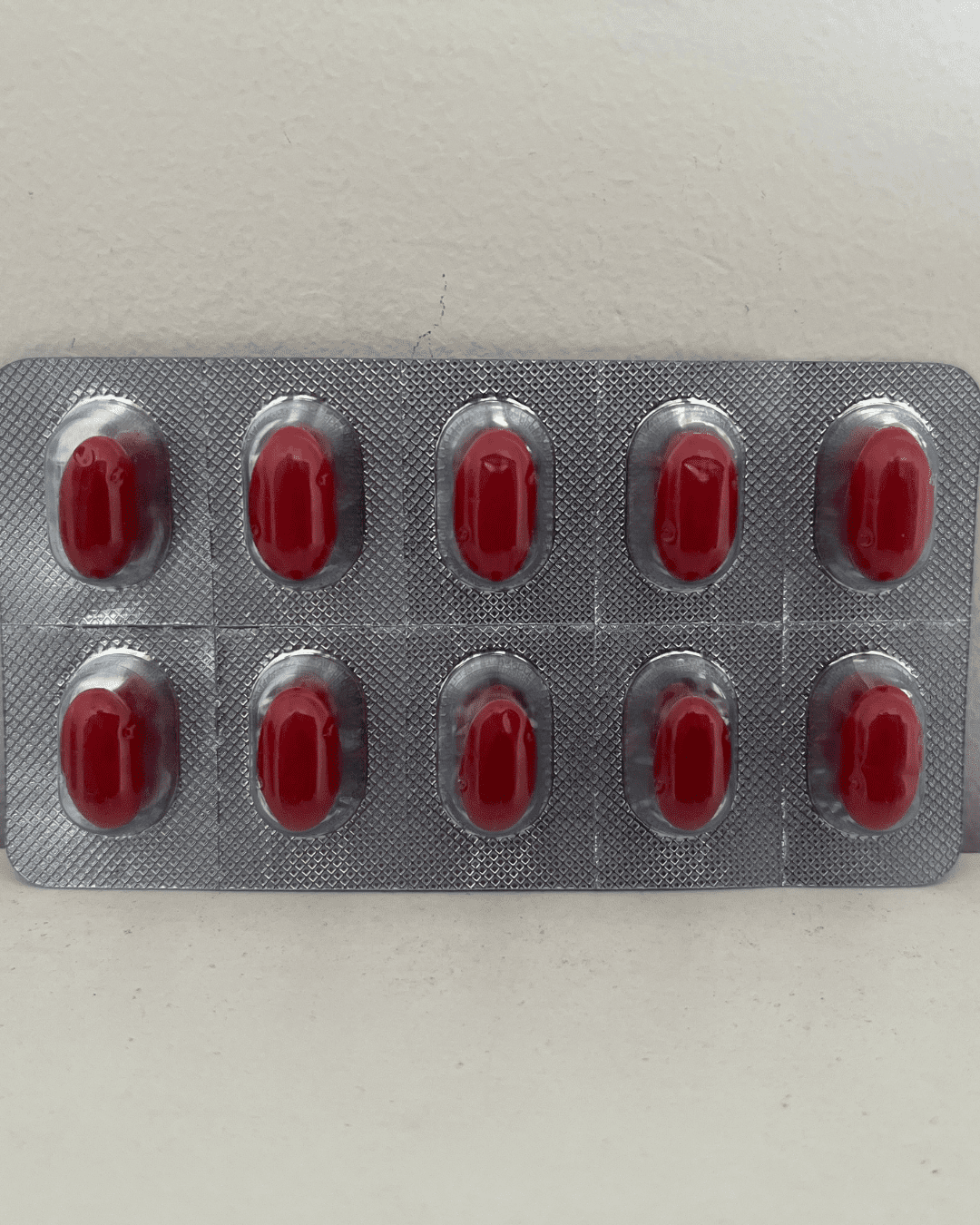
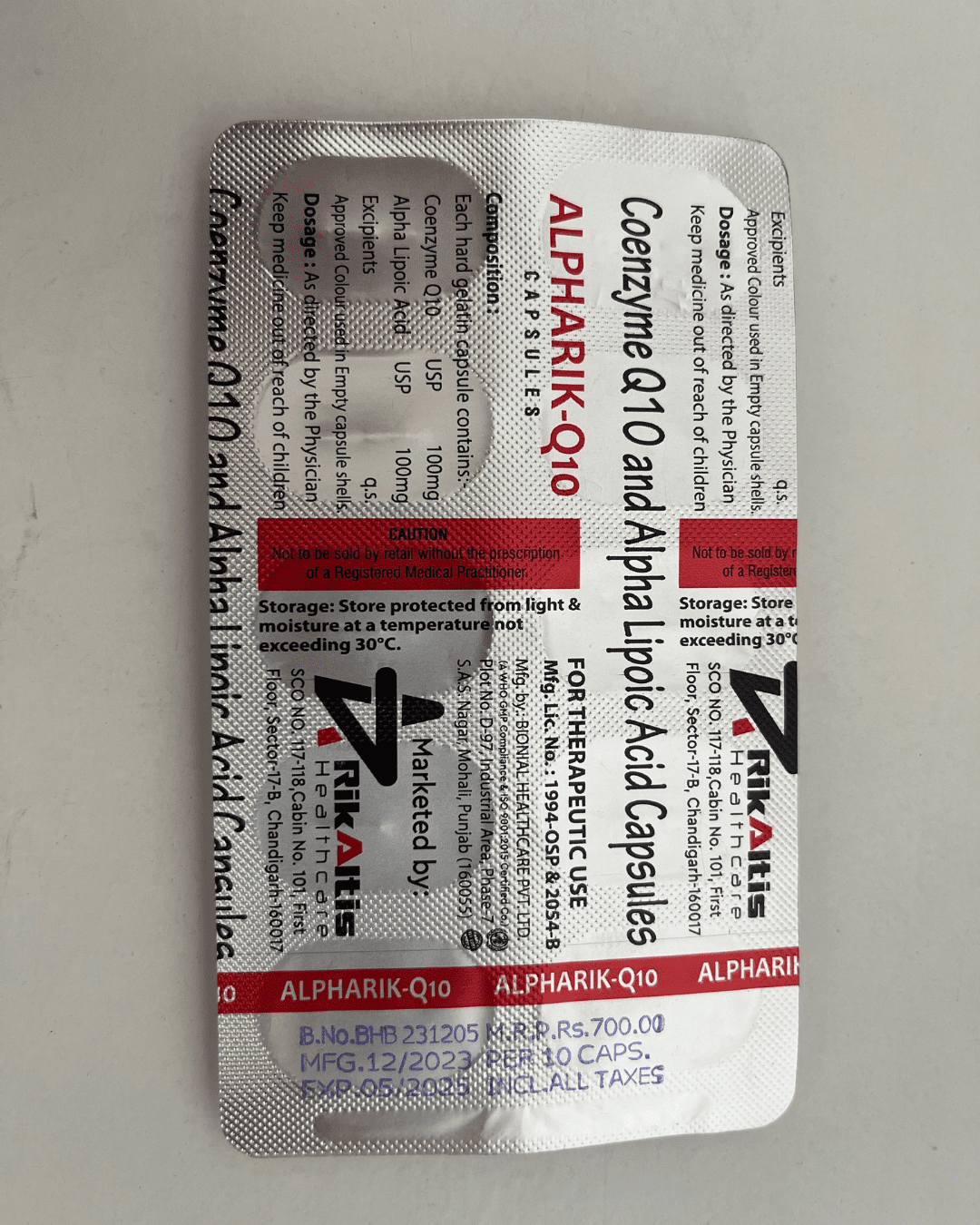
Capsules
|
Premium IV fluids, IVF solutions, and injectable medicines manufactured
with cutting-edge technology and exported to 50+ countries worldwide.
WHO GMP Certified
US FDA Compliant
ISO 14644 Class 100
Leading Manufacturer of
Dry Injections
Liquid Injections
IV Fluids
Tablets
Capsules
|
Premium IV fluids, IVF solutions, and injectable medicines manufactured
with cutting-edge technology and exported to 50+ countries worldwide.
Nuvesa Pharma Pvt. Ltd.
Nuvesa Pharma is a trusted and reputed name in the pharmaceutical industry, specializing in the manufacturing of premium-quality injectable formulations. With a strong vision to enhance global healthcare through innovation and excellence, we are dedicated to delivering safe, effective, and affordable injection solutions that meet the diverse needs of healthcare professionals and patients worldwide.
At Nuvesa Pharma, we operate with a commitment to quality and integrity. Our state-of-the-art manufacturing facility adheres to stringent GMP (Good Manufacturing Practices) and international regulatory standards, ensuring every product meets the highest benchmarks of safety, purity, and efficacy.
Backed by a skilled team of professionals and advanced technology, we focus on the production of sterile injectable products across multiple therapeutic segments.
Let's Get in Touch
Quality Assurance
Committed to the highest standards of pharmaceutical quality through rigorous testing,
international certifications, and continuous improvement

International Certifications
Our facility maintains the highest international standards through comprehensive certifications and regular audits.
WHO GMP
World Health Organization Good Manufacturing Practice
Valid: 2024-2027
Valid
US FDA
United States Food and Drug
Administration
Valid: Current
Valid
ISO 14644
Cleanroom Classification Class 100
Valid: 2024-2025
Valid
CE Marking
European Conformity Declaration
Valid: Current
Valid
ISO 9001:2015
Quality Management System
Valid: 2023-2026
Valid
ISO 13485
Medical Devices Quality Management
Valid: 2024-2027
Valid

Advanced Packaging Solutions
Our state-of-the-art packaging technologies ensure product integrity, sterility, and compliance with international standards.
Euro Head
European standard packaging
BFS Technology
Blow-Fill-Seal sterile packaging
Glass Vials
Premium borosilicate glass
Plastic Containers
Medical grade materials
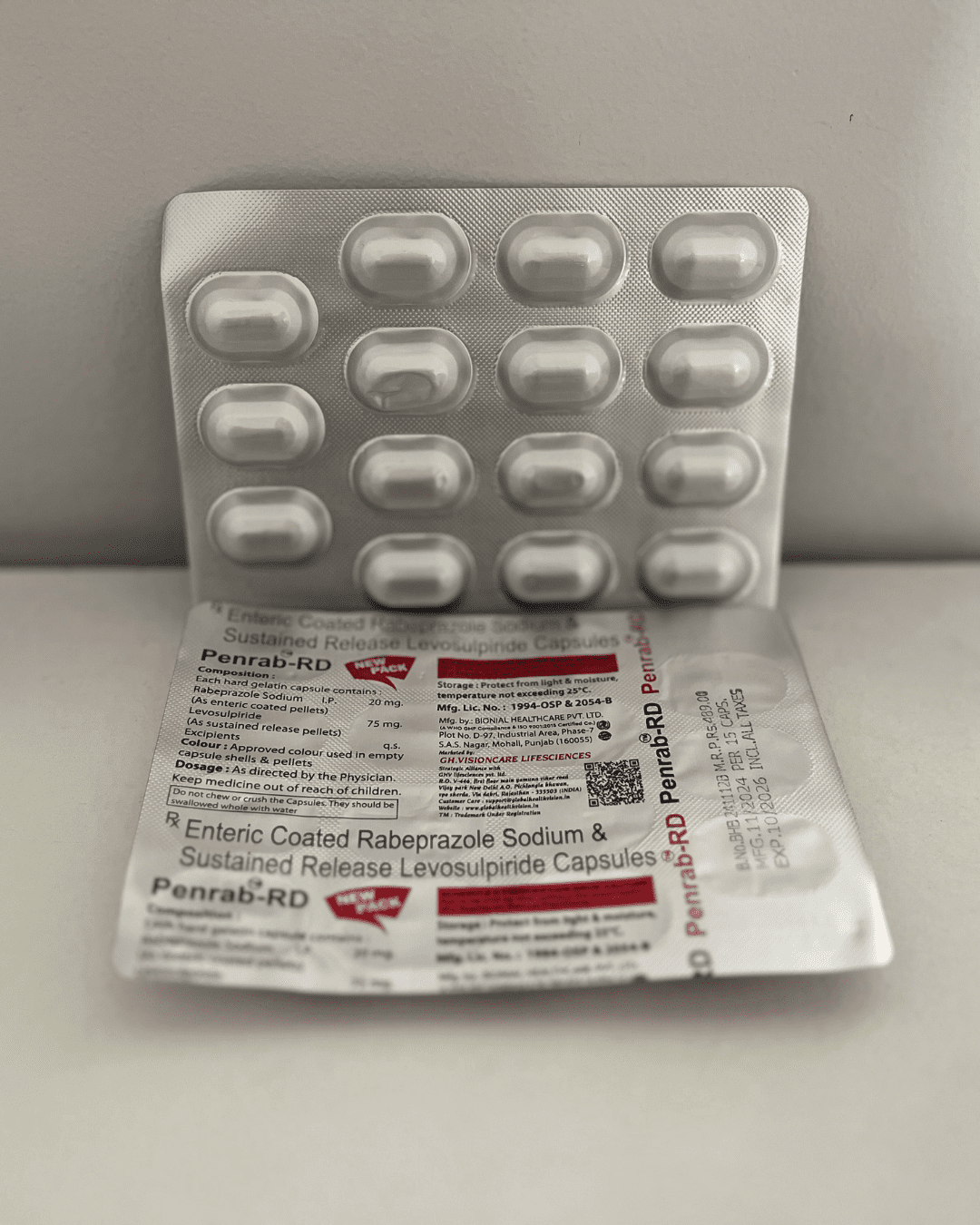
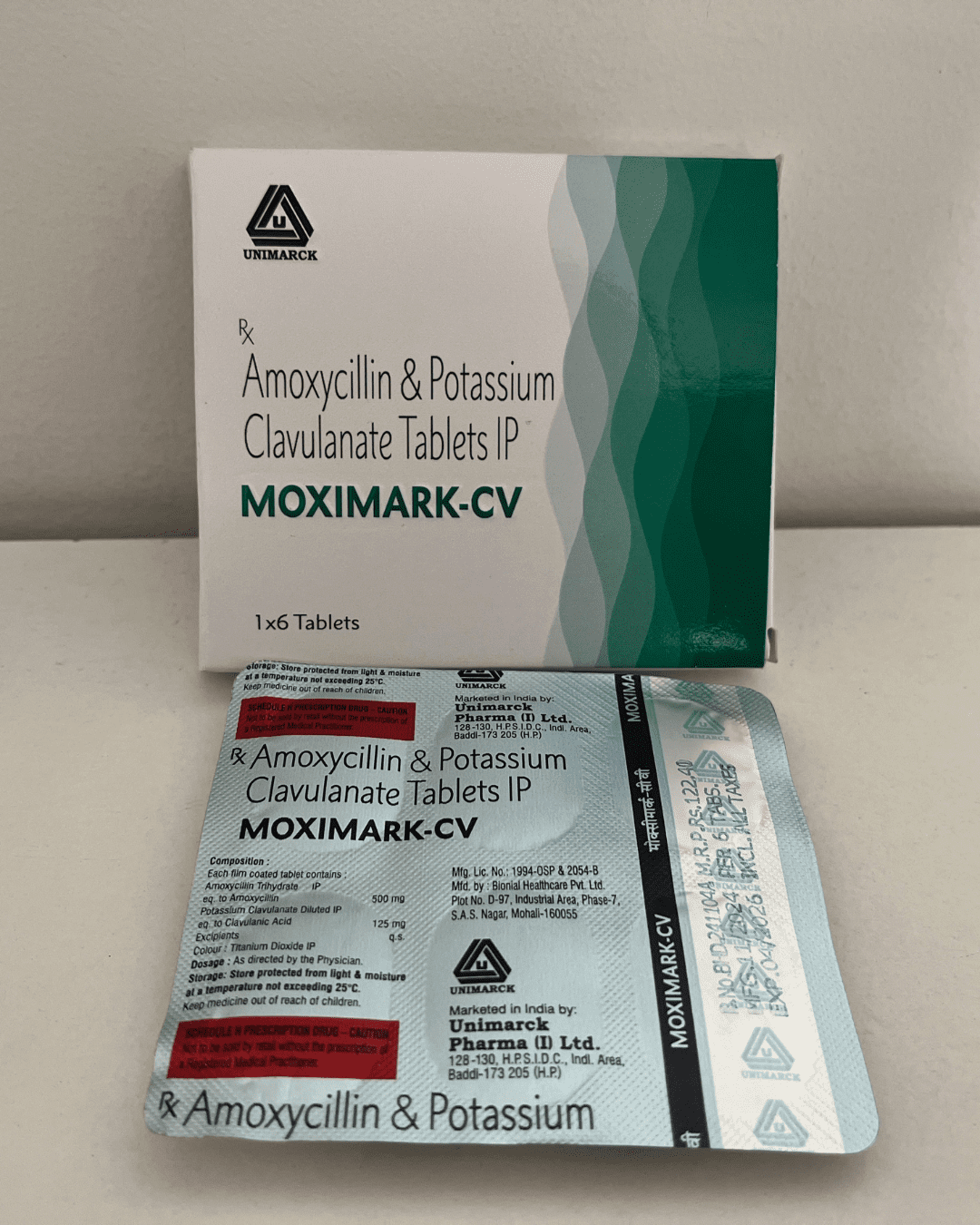
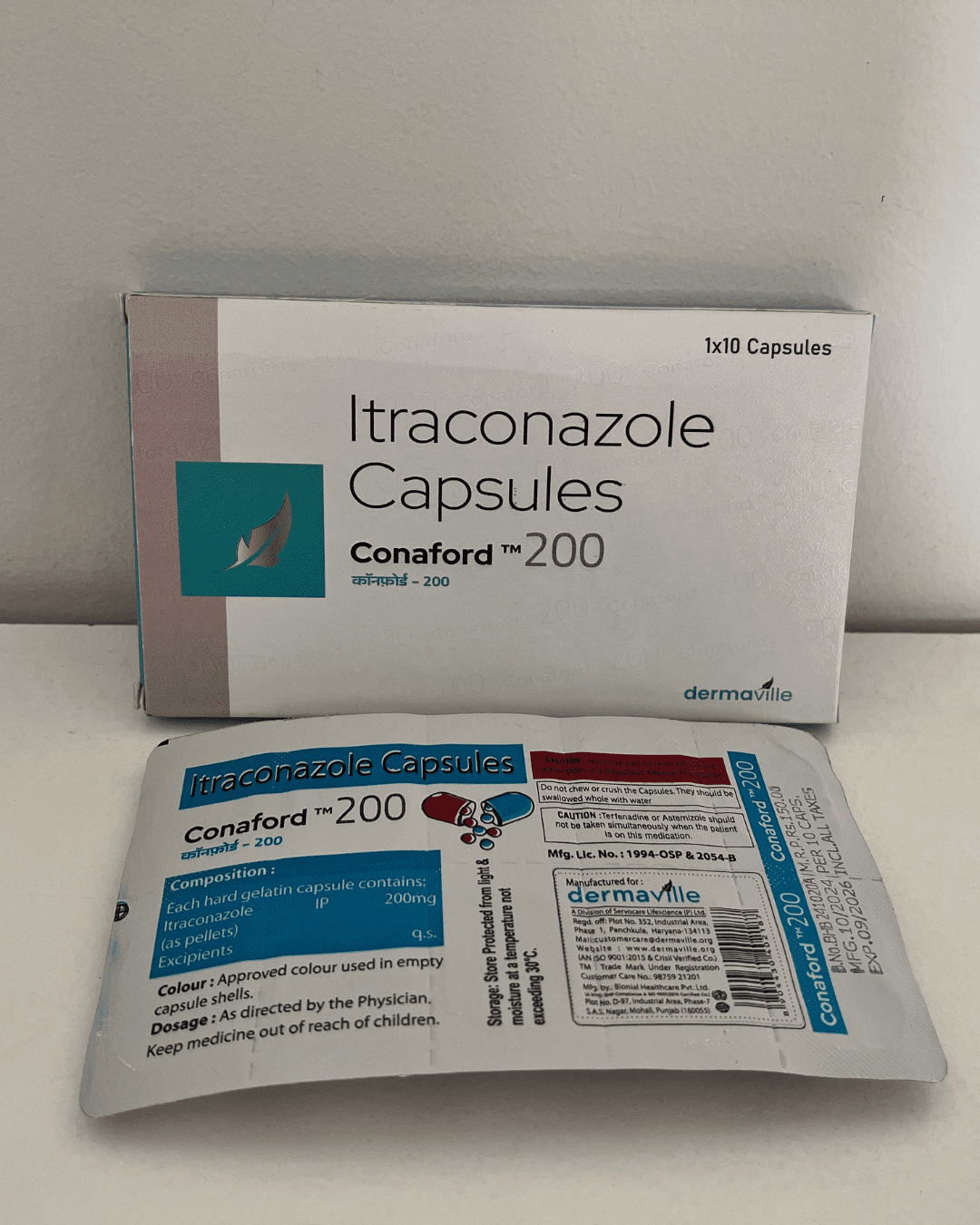
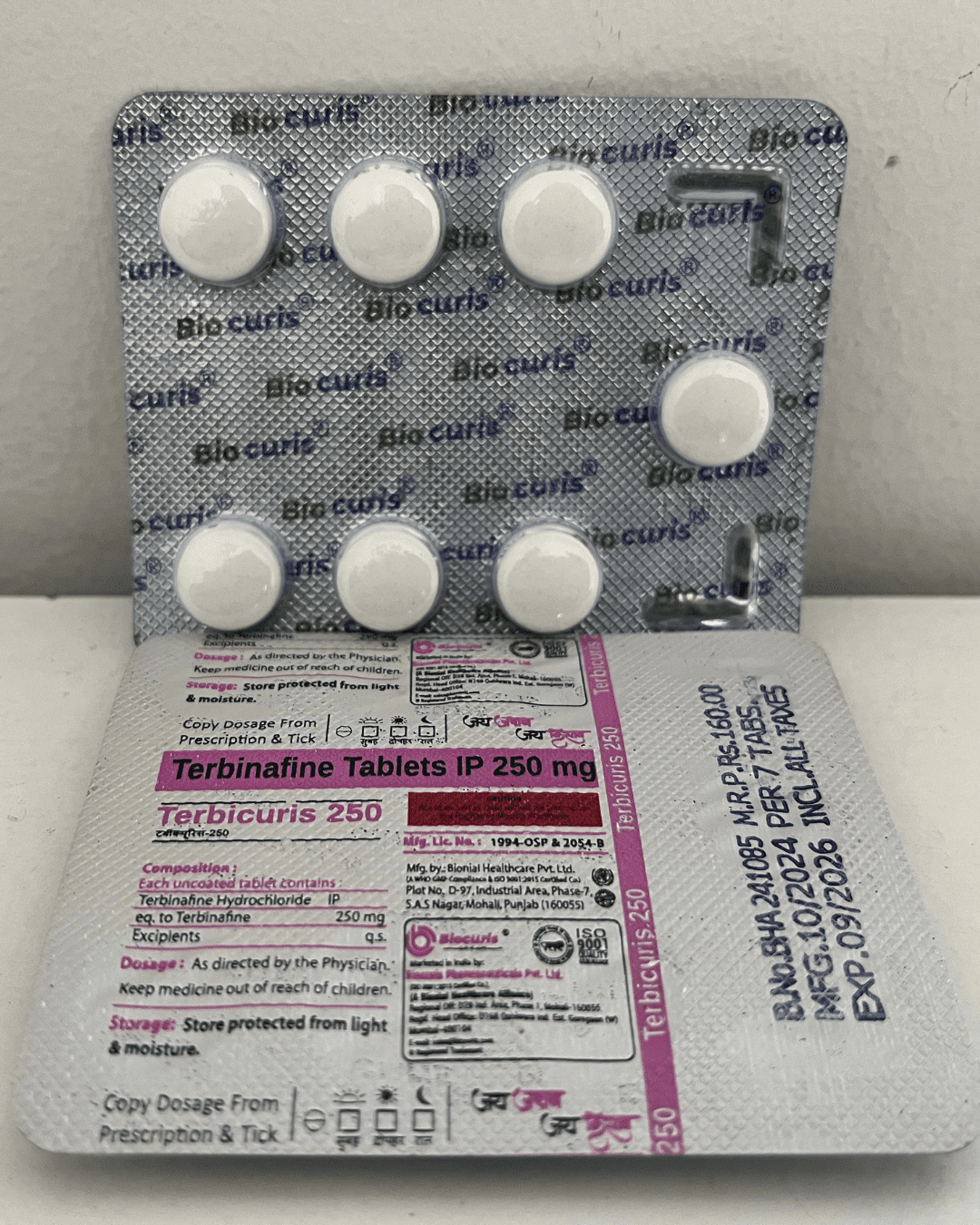
Our Products Range
What Our Client Says
"Nuvesa Pharma has been a reliable partner for us over the years. Their injectable formulations consistently meet global quality standards, and their commitment to timely delivery and customer support is commendable."
– Meera S.
"As a healthcare practitioner, I value consistency and safety in the medicines I prescribe. Nuvesa Pharma’s injectables have always delivered reliable results and meet the highest standards of purity and efficacy."
– Rahul D.
"Working with Nuvesa Pharma has been a seamless experience. Their transparent communication, GMP-certified facilities, and focus on quality assurance make them one of the best pharma partners we’ve worked with."
– Aarti T.
"We source several sterile injectable products from Nuvesa Pharma, and their quality, packaging, and documentation always meet international expectations. Their professionalism and responsiveness set them apart."
– Dr. Kunal M.
"Nuvesa Pharma truly understands market needs. Their product portfolio is diverse, and their support team ensures we always receive our orders on time. A trustworthy brand in the injectable pharma segment."
– Renu V.
"Nuvesa Pharma has been a reliable partner for us over the years. Their injectable formulations consistently meet global quality standards, and their commitment to timely delivery and customer support is commendable."
– Meera S.
"As a healthcare practitioner, I value consistency and safety in the medicines I prescribe. Nuvesa Pharma’s injectables have always delivered reliable results and meet the highest standards of purity and efficacy."
– Rahul D.
"Working with Nuvesa Pharma has been a seamless experience. Their transparent communication, GMP-certified facilities, and focus on quality assurance make them one of the best pharma partners we’ve worked with."
– Aarti T.
"We source several sterile injectable products from Nuvesa Pharma, and their quality, packaging, and documentation always meet international expectations. Their professionalism and responsiveness set them apart."
– Dr. Kunal M.
"Nuvesa Pharma truly understands market needs. Their product portfolio is diverse, and their support team ensures we always receive our orders on time. A trustworthy brand in the injectable pharma segment."
– Renu V.
Quality Control Process

Raw Material Testing
Comprehensive analysis of all incoming materials

In-Process Monitoring
Real-time quality control during production

Finished Product Testing
Complete validation before release

Stability Studies
Long-term product stability validation

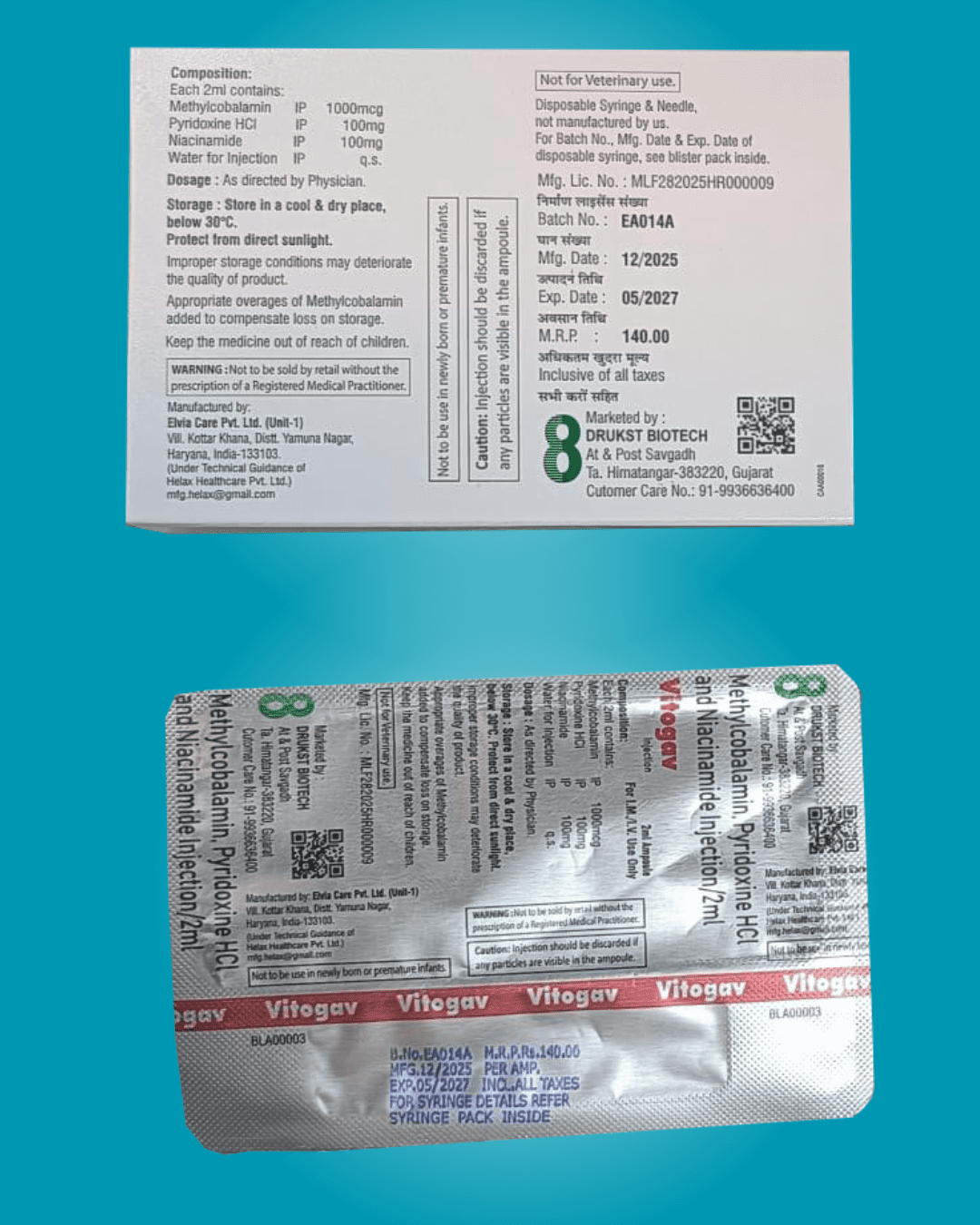

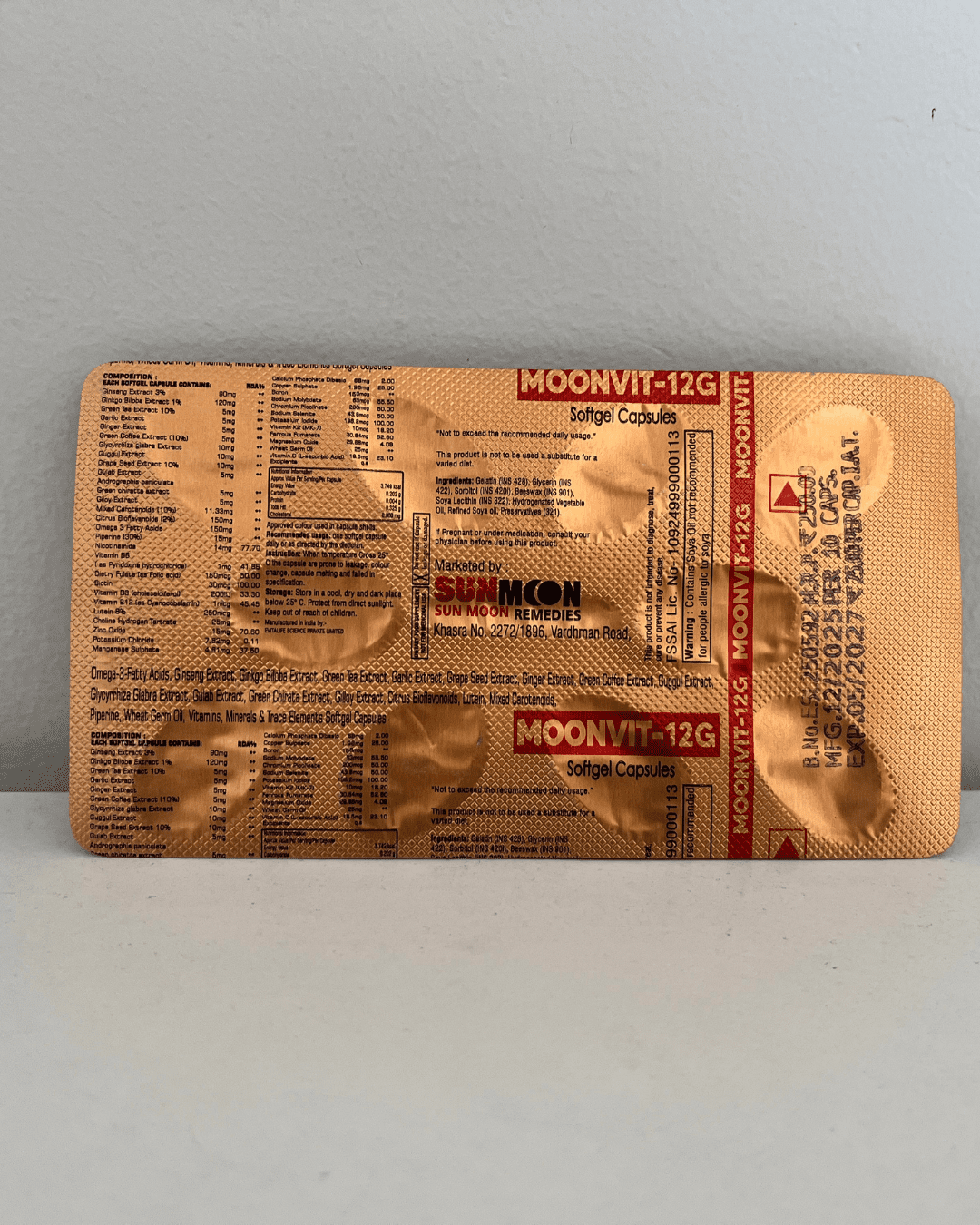






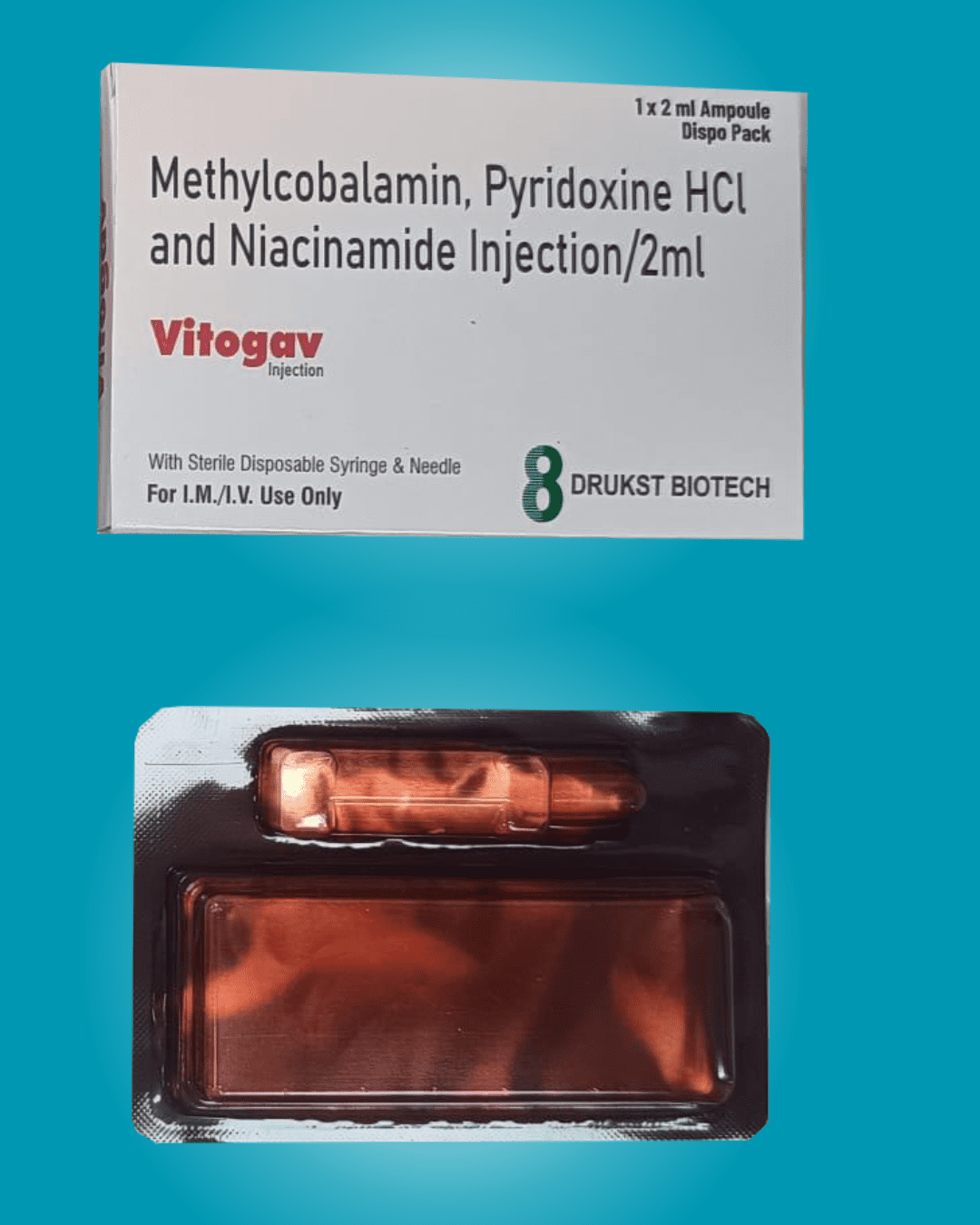

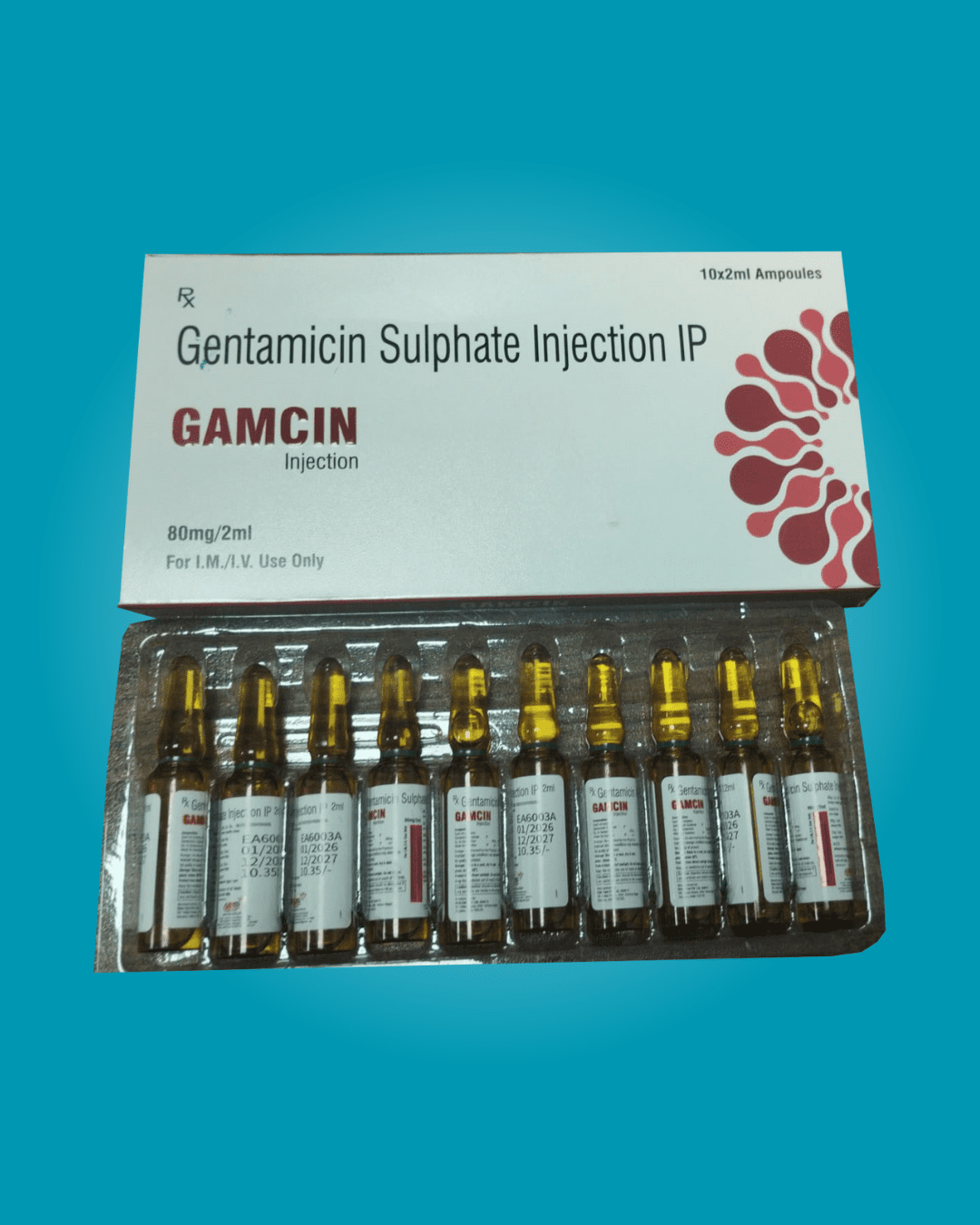







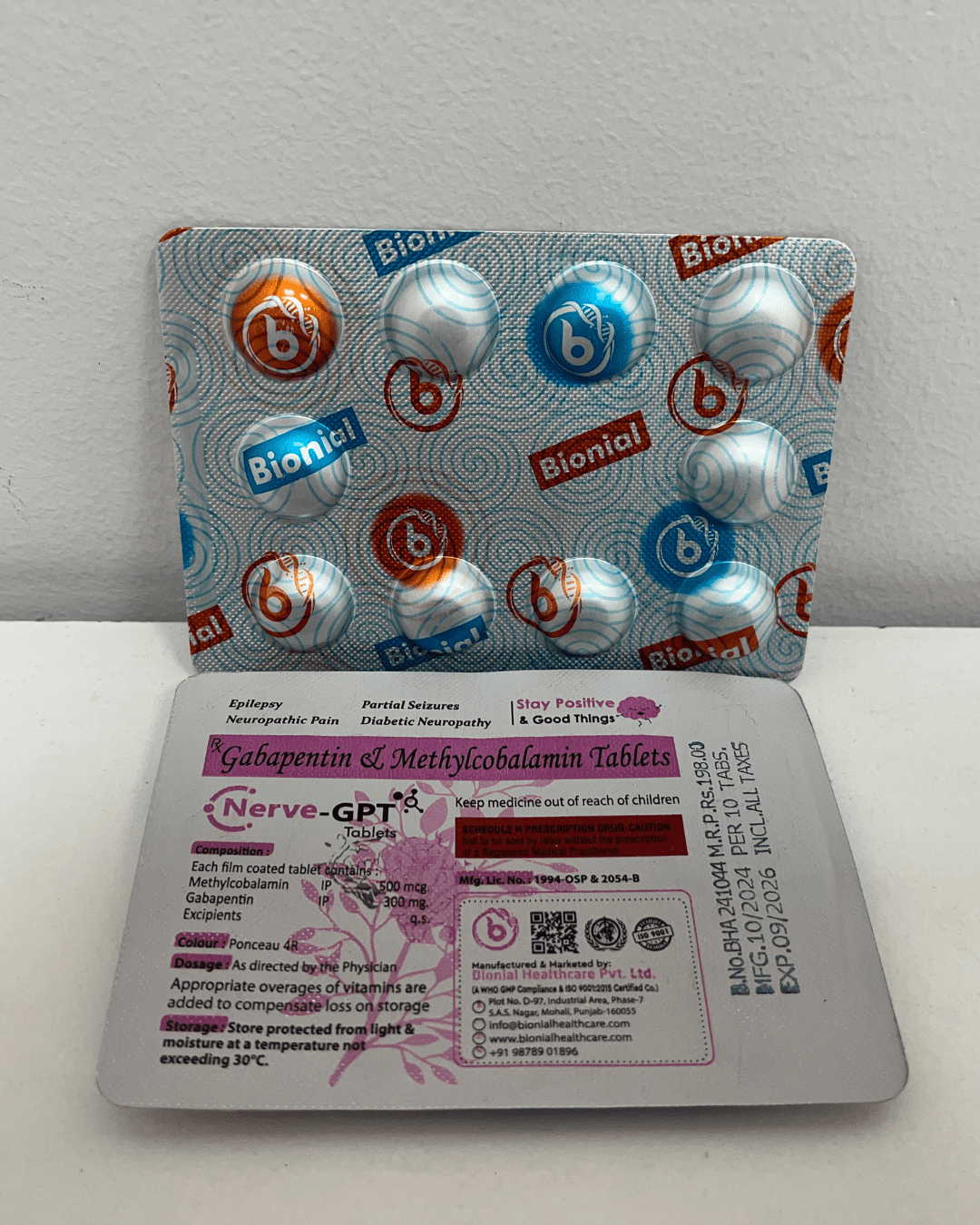
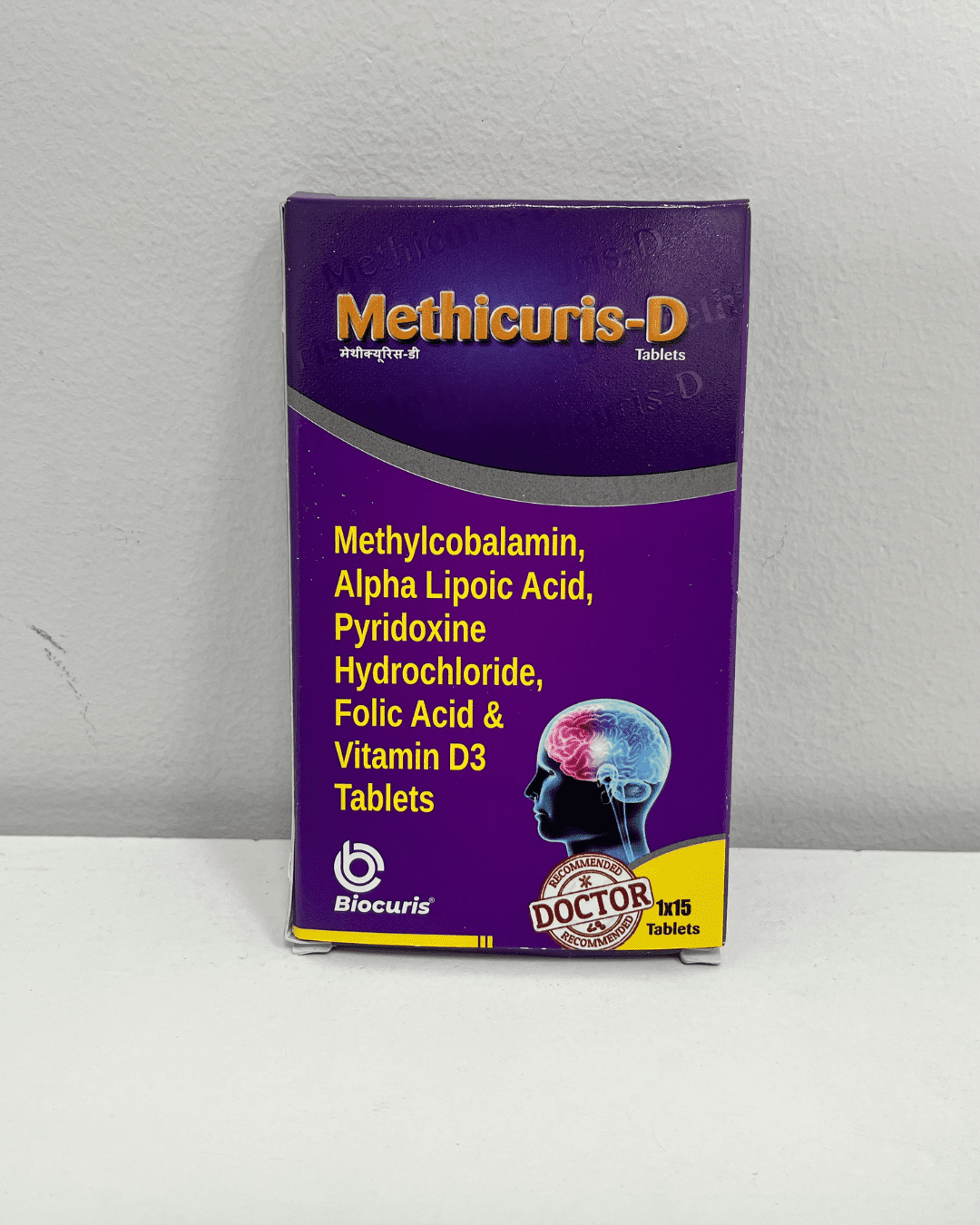


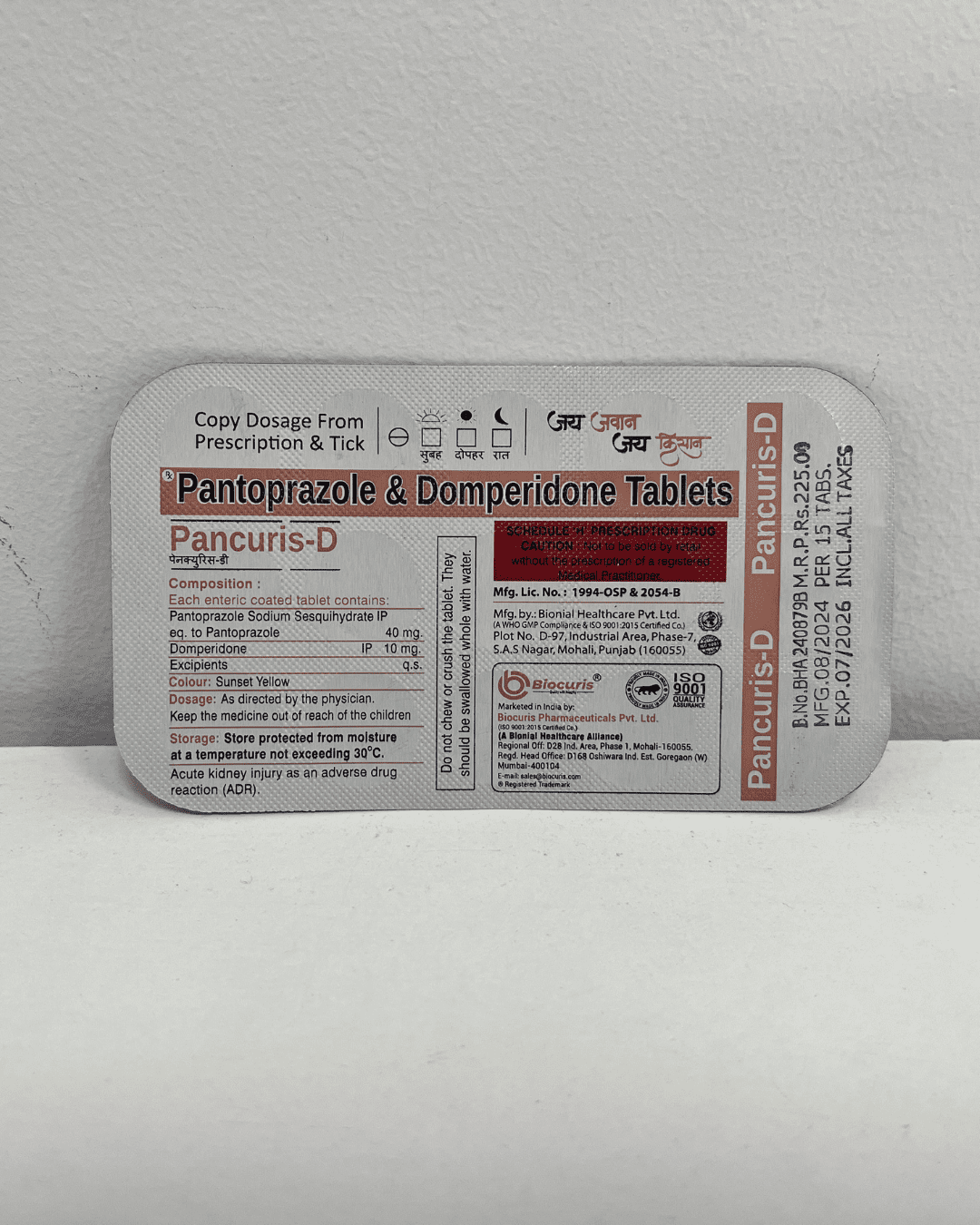

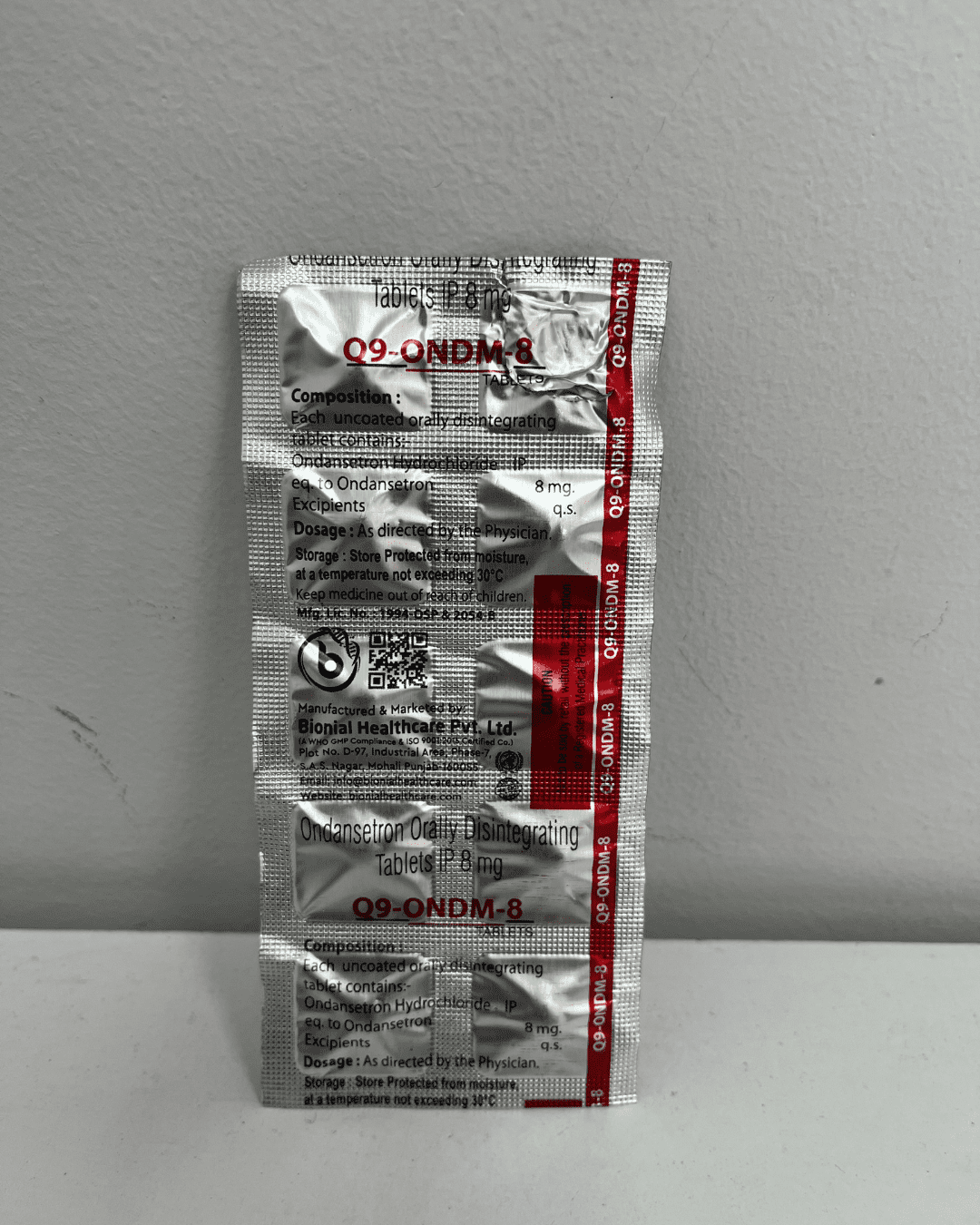
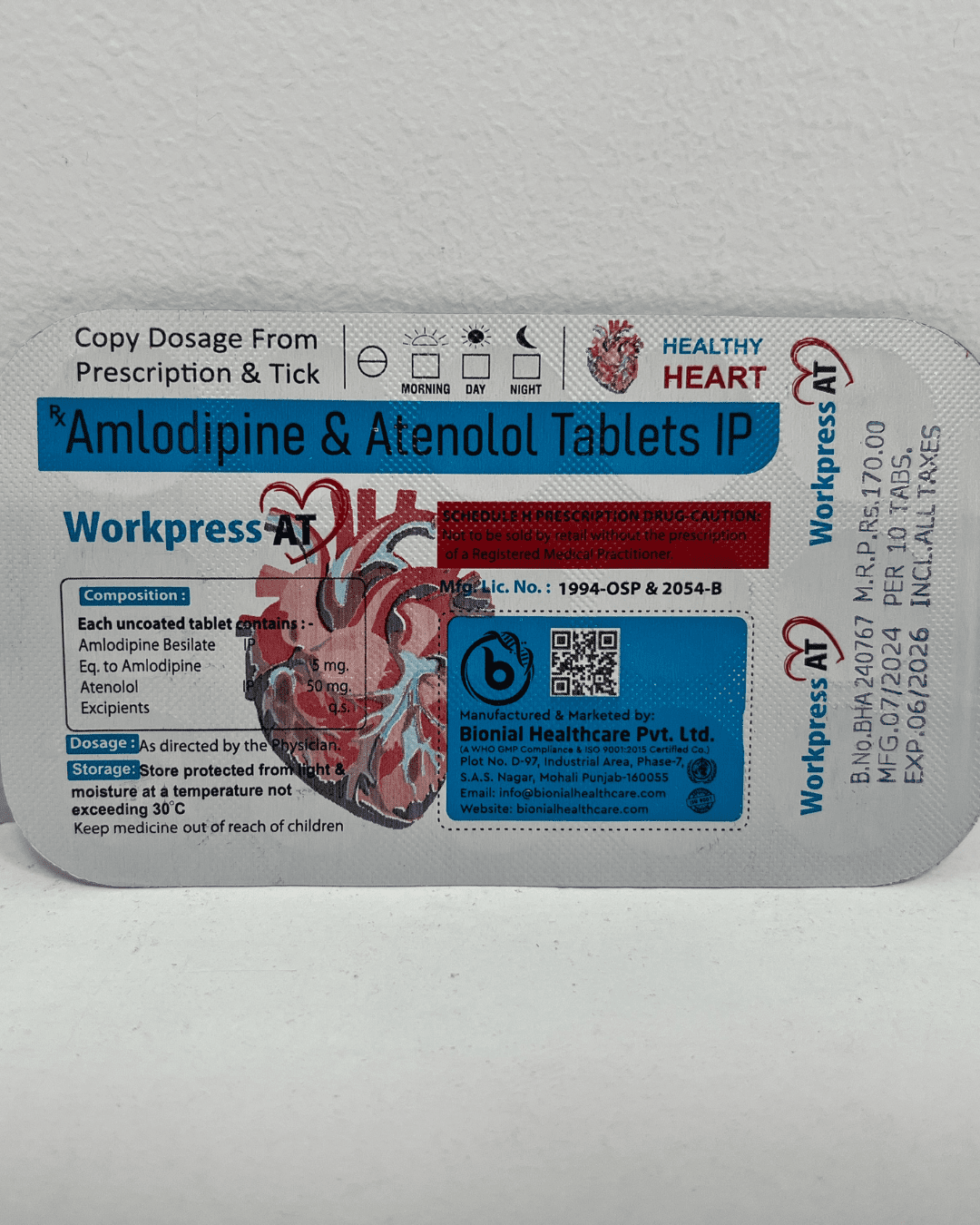
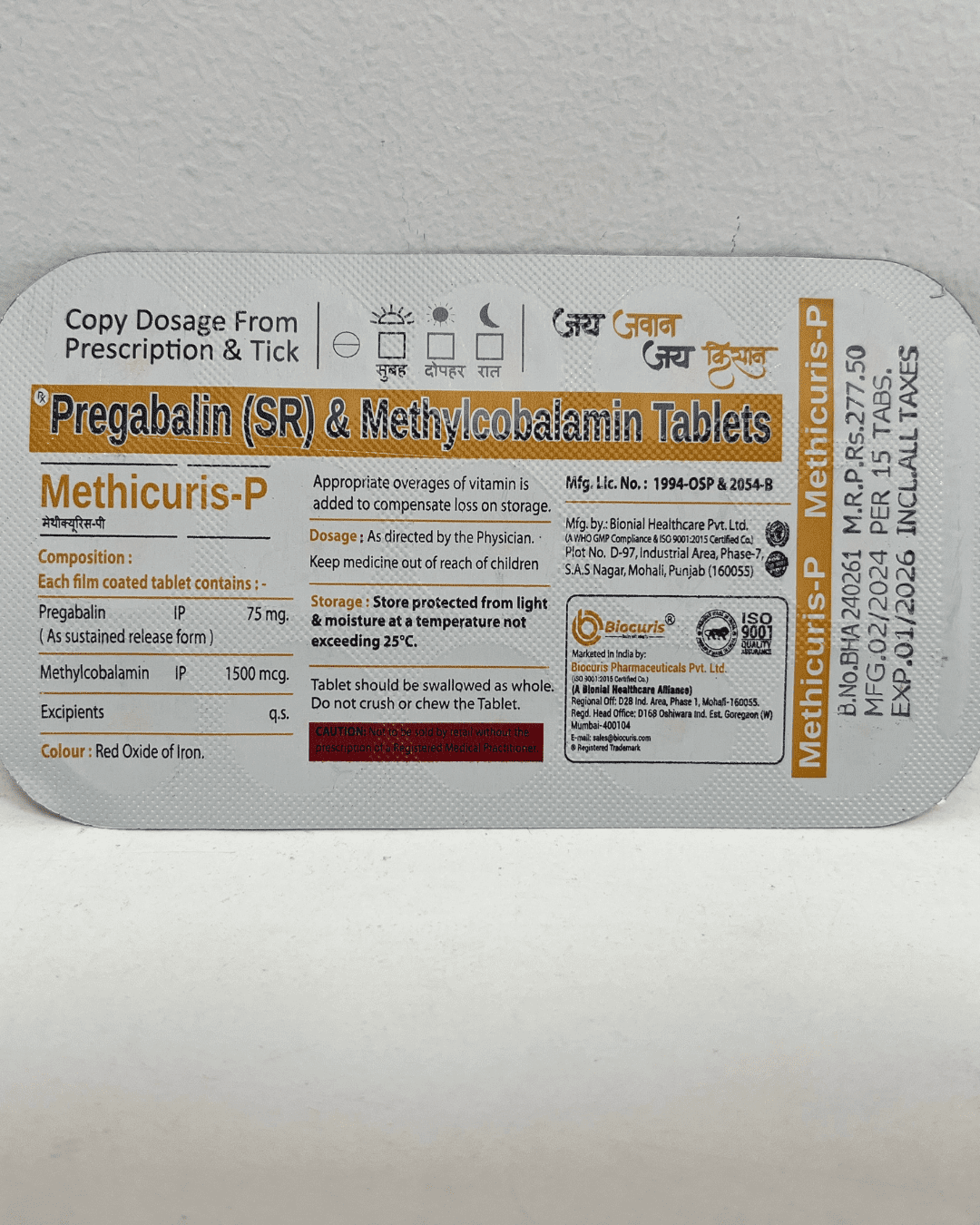

Regulatory Compliance
Our products meet the stringent requirements of global regulatory bodies
ICH Guidelines
USP Standards
European Pharmacopoeia
WHO Technical Reports
Let's connect

Global Headquarters
SCO 57-A & 58, Second floor, Chandigarh City Center, Vip Road, Zirakpur, Punjab 140603

Export Department
export@nuvesapharma.com
+91 9056072355
24/7 Support Available

Domestic Business Unit
bd@nuvesapharma.com
+91 9056072355
Regulatory Compliance




